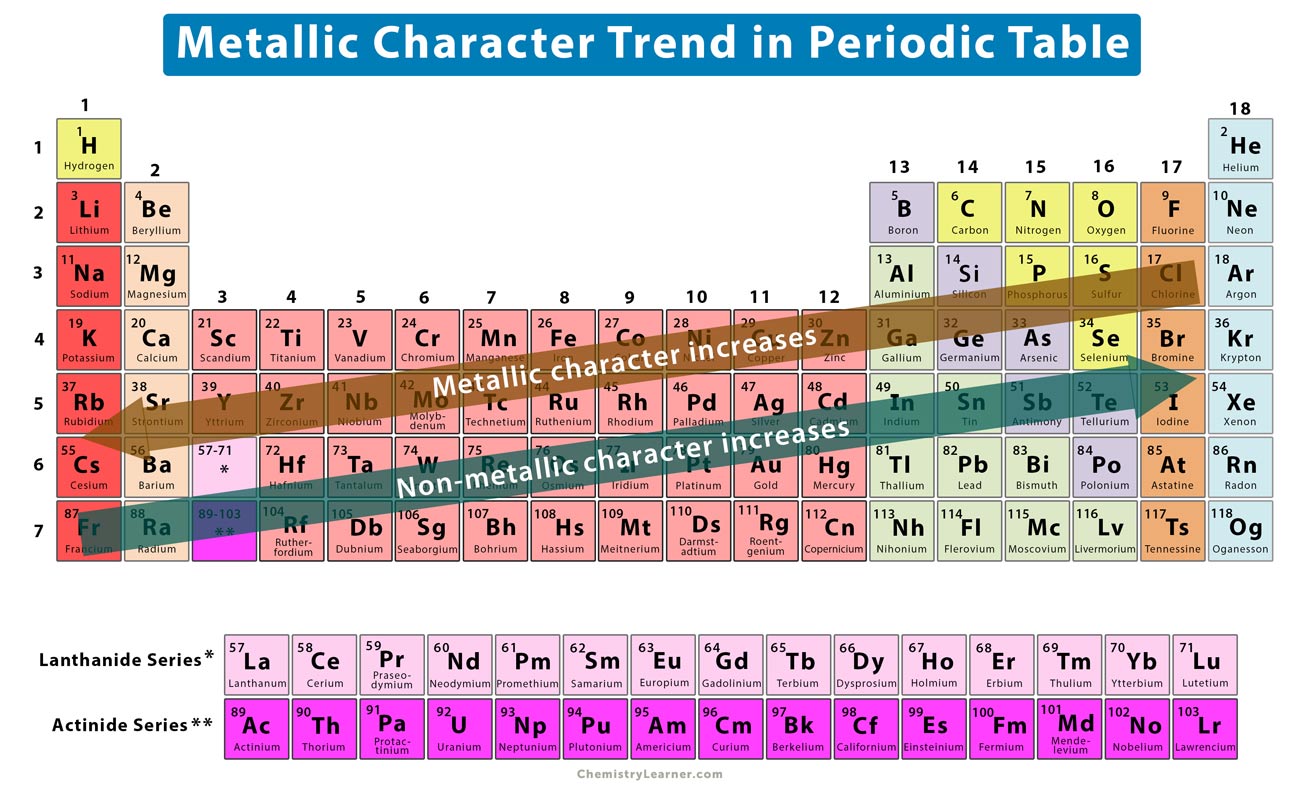
For example, a quick glance at Figure AT5.2. That way, we can get a better look at the relationship. Often it is useful to plot data on a graph. As a result, fluorine is usually thought of as the most electronegative element. However, on many scales, fluorine would be the most electronegative atom here. Non-metallic tendency increases going from left to right across the periodic table. Metallic tendency increases going down a group. Non-metallic character relates to the tendency to accept electrons during chemical reactions. As a result, noble gases are also given electronegativity values on this scale. Metallic character refers to the level of reactivity of a metal. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. The elements on the left tend to be more reactive and the elements on the right tend to be less reactive.Ĩ.) Arrange each of the following metals in order from most to least reactive.\): The Allen electronegativity values of the second-row elements. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends. As you move higher up a column, the elements tend to be more reactive.ħ.) What can you conclude about the reactivity of metals as you move across a period? Some trends I learned from these labs are that, starting from left to right, the elements on the left are more reactive and the elements on the right are less reactive.Ħ.) What can you conclude about the reactivity of metals as you move down a column or group in the periodic table? 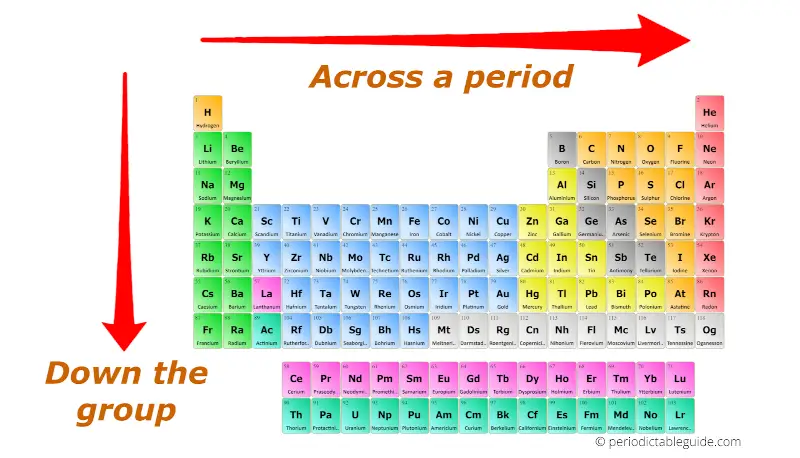 
Describe some of the trends that you learned about from this lab? based on this trend, what do you estimate the density of silicon to be?ĥ.) When elements are organized in the periodic table, various trends appear. The density of tin is about 7.28g/cm ³ and the density of Pb is 11.34 g/cm ³. Elements in the periods near the top of the periodic table tend to react more, while those close to the metalloid boundary and in the bottom left do not react as much.Ĥ.) Silicon, tin, and lead are all in the same group. The repeating properties in each row of the periodic table, as observed by Mendeleev and others, reflect the repeating electron configurations in subsequent rows. Use data from your lab to support your answer.ģ.) In general, is there a relationship between the locations of metals on the periodic table and their relative activity? Also, magnesium and calcium are in different periods on the periodic table, which may indicate that they react differently.Ģ.) List the four metals from most reactive to least reactive. The difference in behavior might have been because magnesium was in a solid form and the calcium was dehydrated and powdered, which appeared to mix with the water easily and dissolve. Wash test tubes.ġ.) What might be a reason for the difference in behavior between magnesium and calcium placed in water? Procedure - Part 1: Trends in Properties Within GroupsĤ.) Fill a beaker with tap water and get a disposable pipette.ĥ.) Bring test tubes to Charlene to get a sample of magnesium and a sample of calcium.Ħ.) Use pipette to cover sample with water.ħ.) Observe reaction carefully and enter data in table.Ĩ.) Dump sample into hand in sink, rinse sample, and place in corresponding labeled beaker. Periodic trends (such as electronegativity, electron affinity, atomic and ionic radii, and ionization energy) can be understood in terms of Coulombs law, which is F (qq)/r². IE decreases bc the atomic radius increases, valence electrons dont feel the pull of the. the amount of energy required to remove an electron. negative ion formed by gaining electrons, atomic radius increases. Gallium is in Group 3 and the 3rd period. positive ion formed by losing electrons, atomic radius decreases. The alkali metals are the first group on the periodic table and the alkali earth metals are the second group.Ĥ.) What element is in Group 3 and the 3rd Period? The metals are located to the left of the metalloid boundary.ģ.) What are the names of two metal families? The groups of the periodic table that share a lot of similarities with all of the other elements in that group are called families.Ģ.) In general, where are the metals located on the periodic table? The periods of the periodic table are the horizontal rows. Groups of the periodic table are the vertical columns. To explore the reactivity trends of metals in groups and periods of the periodic table.ġ.) What are groups? What are periods? What are families?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
